Supreme Info About How To Draw A Covalent Bond

Arrange the atoms so that the valence electrons will complete the outer shells of all the atoms in a simple.
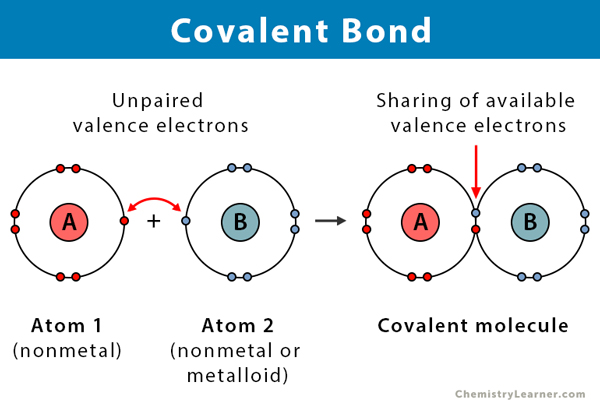
How to draw a covalent bond. Can the control room and power distribution room be built in the class a workshop? A covalent bond is a chemical bond that comes from the sharing of one or more electron pairs between two atoms. A covalent bond is when an atom shares their electron with another atom.
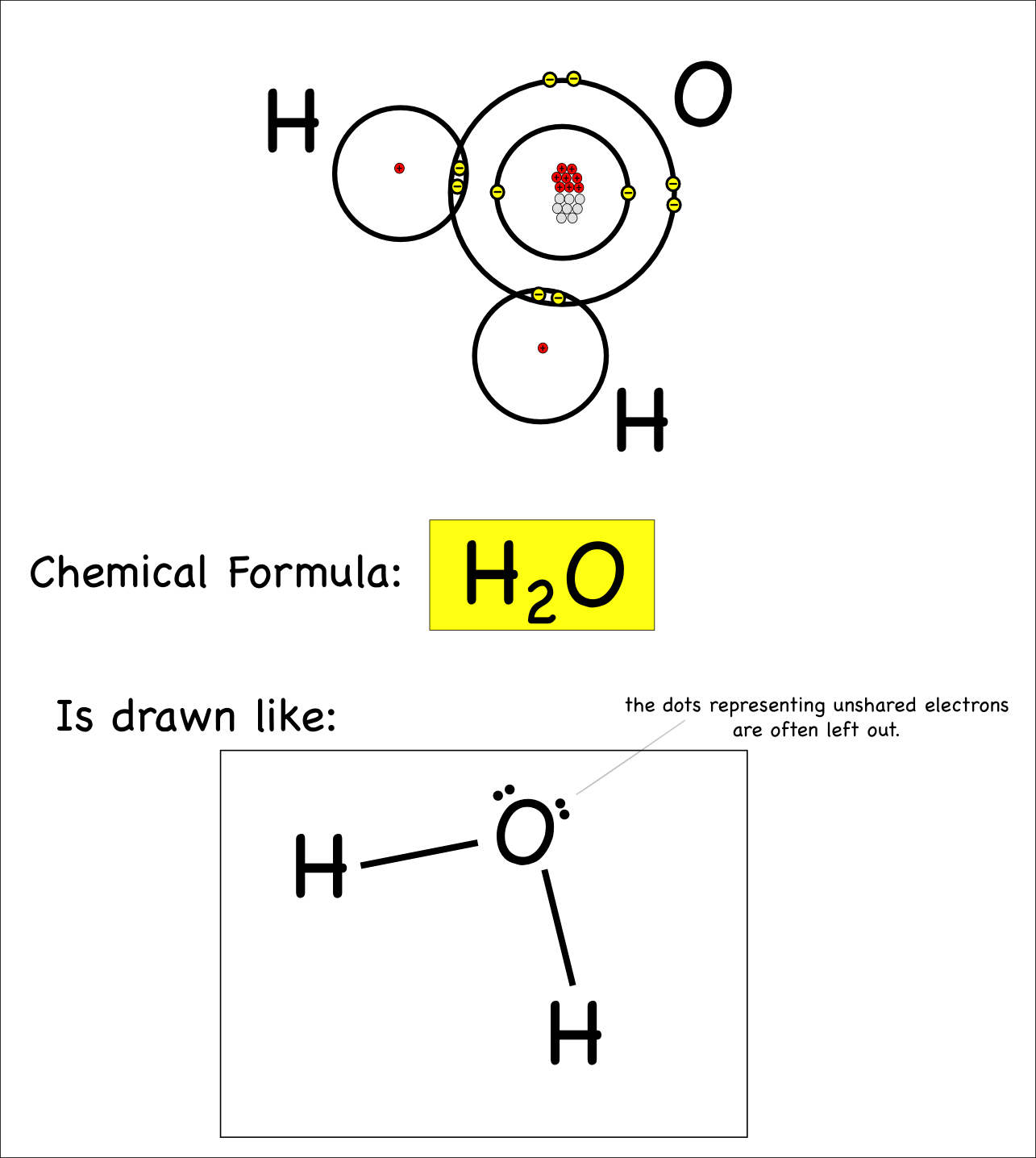
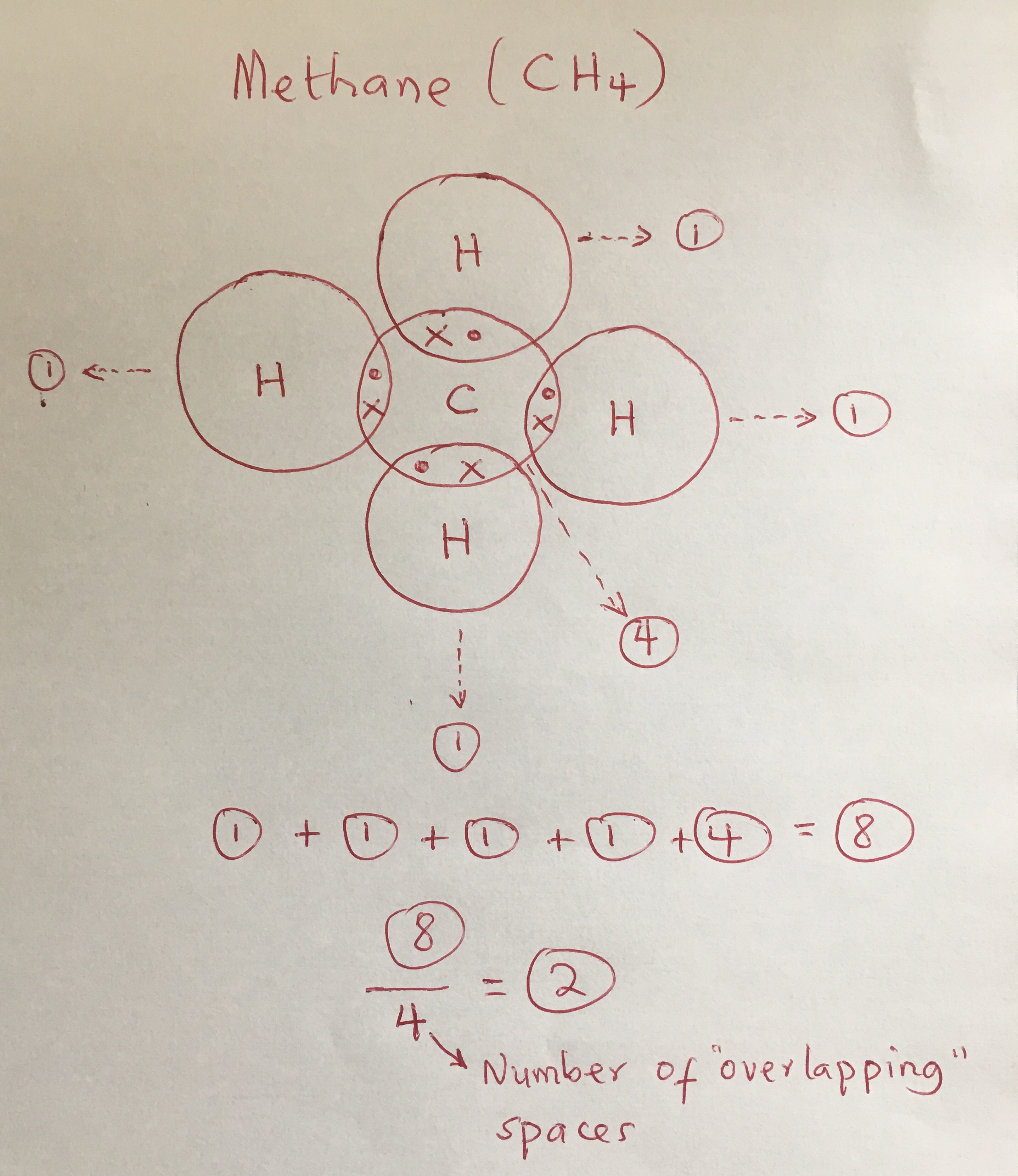
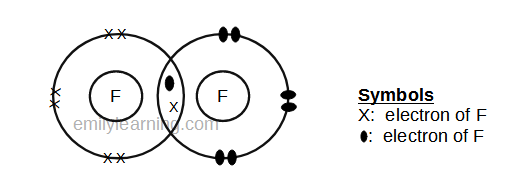
As covalent bonds are formed by sharing of electrons between atoms, we draw overlapping circles to show the overlapping of the electron shells, and draw in pairs of dots. There are different kinds of covalent bonds: You don't need to put a circle around the symbol for the nucleus.
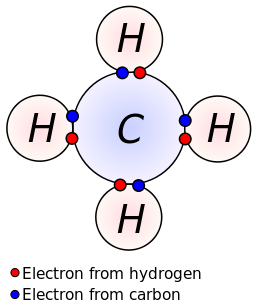
The sharing of electrons between atoms of different elements (for example h2o, ch4 and nh3 etc). About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Each of the three h circles overlaps the n circle.
In this simpler type of diagram, the atoms are represented by writing their chemical symbols and a single covalent bond is represented by drawing a straight line between the two atoms. For this, draw four circles, one labelled n and three labelled h. The two ways in which covalent bonding can be achieved:
Draw a lewis dot structure for the valence shell of each element. How to draw a lewis structure. It depends on the type of element that you're drawing it for.
Practice covalent compounds (molecular compounds):• h₂o water (dihydrogen monoxide)• o₂ oxygen• co₂ carbon dioxide• hf hydrogen fluoride• ch₄ methane• p. It is often easiest to draw circles at 90° or 180° to each other nitrogen is. Contents:0:08 introduction 0:39 h21:25 hcl2:23 cl2.
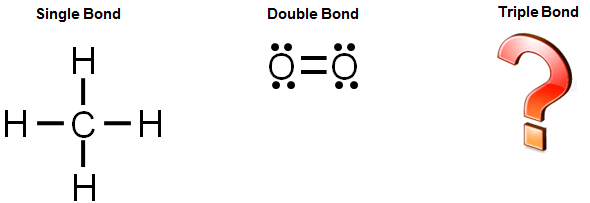
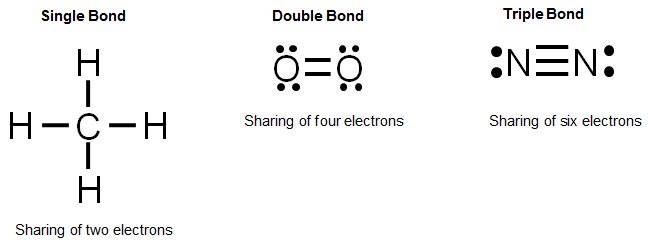
To determine how many leftovers. A single covalent bond is when two atoms share a single pair of electrons. How do you draw a dot diagram for a covalent compound?
Given descriptions, diagrams, scenarios, or chemical symbols, students will model covalent bonds using electron dot formula (lewis structures). In a lewis structure, atoms that are bonded covalently are. Each pair of shared electrons is a covalent bond which can be represented by a dash.
In this type of formula, valence. Find the total number of valence electrons.